|
|
Inspiration
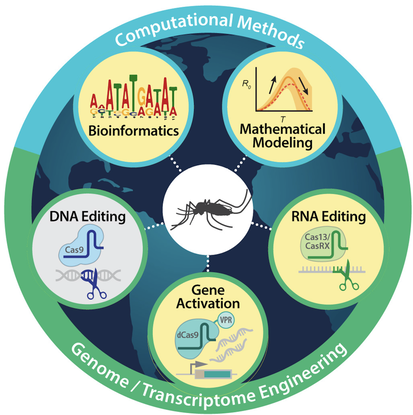
Mosquitoes are perhaps the most dangerous animals in the world. They are the primary vectors for major human diseases such as yellow fever, zika, dengue fever, and malaria, which together infect hundreds of millions of humans worldwide, killing millions each year, with over 50% of the world’s population presently at risk (WHO). There are currently no effective vaccines for many mosquito vectored pathogens, and mosquitoes, including the disease-causing agents they transmit, are rapidly evolving resistance to commonly used pesticides and drugs. Therefore, given the number of infections and deaths, current approaches for prevention of mosquito-borne diseases are immeasurably inefficient. What remains critical for vector control is the development of catalytic approaches requiring minimal efforts that can generate long lasting solutions.
With rapid advances in insect genetic engineering, mathematical modeling of wild populations, synthetic biology, and the comprehensive understanding of pathogen lifecycles in mosquitoes, unique opportunities have arisen to eliminate infectious diseases through genetic manipulation of wild insect vector populations. Taking advantage of these opportunities our research focuses on studying the basic genetics and physiology of mosquitoes with the overarching goal of developing innovative, novel, creative, synthetic biology inspired genetic control technologies for reducing the burden of mosquito-borne diseases on humans. The underlying hypothesis inspiring this work is that the introduction and spread of genes that prevent mosquitoes from transmitting pathogens should, in theory, lead to reduced transmission of these pathogens resulting in reductions of human infections and/or death. To test this hypothesis, we first need a broad understanding of the biology of the mosquito that can be used to develop gene-based strategies for engineering mosquitoes that are resistant to pathogens; secondly, we need to engineer mosquitoes that are resistant to all types of infections; and lastly, we need to develop tools to rapidly “drive” these laboratory developed genes into wild mosquito populations. Together, these aims can conceivably provide a foundation that has the potential to revolutionize vector control of mosquitoes.
With rapid advances in insect genetic engineering, mathematical modeling of wild populations, synthetic biology, and the comprehensive understanding of pathogen lifecycles in mosquitoes, unique opportunities have arisen to eliminate infectious diseases through genetic manipulation of wild insect vector populations. Taking advantage of these opportunities our research focuses on studying the basic genetics and physiology of mosquitoes with the overarching goal of developing innovative, novel, creative, synthetic biology inspired genetic control technologies for reducing the burden of mosquito-borne diseases on humans. The underlying hypothesis inspiring this work is that the introduction and spread of genes that prevent mosquitoes from transmitting pathogens should, in theory, lead to reduced transmission of these pathogens resulting in reductions of human infections and/or death. To test this hypothesis, we first need a broad understanding of the biology of the mosquito that can be used to develop gene-based strategies for engineering mosquitoes that are resistant to pathogens; secondly, we need to engineer mosquitoes that are resistant to all types of infections; and lastly, we need to develop tools to rapidly “drive” these laboratory developed genes into wild mosquito populations. Together, these aims can conceivably provide a foundation that has the potential to revolutionize vector control of mosquitoes.

