|
1 Comment
WHY DO WE NEED TO DO FIELD RELEASES OF GENE DRIVE TECHNOLOGIES AS PART OF THE R&D PROCESS?10/5/2022 WHY DO WE NEED TO DO FIELD RELEASES OF GENE DRIVE TECHNOLOGIES AS PART OF THE R&D PROCESS? https://genedrivenetwork.org/blog/256-why-do-we-need-to-do-field-releases-of-gene-drive-technologies-as-part-of-the-r-d-process
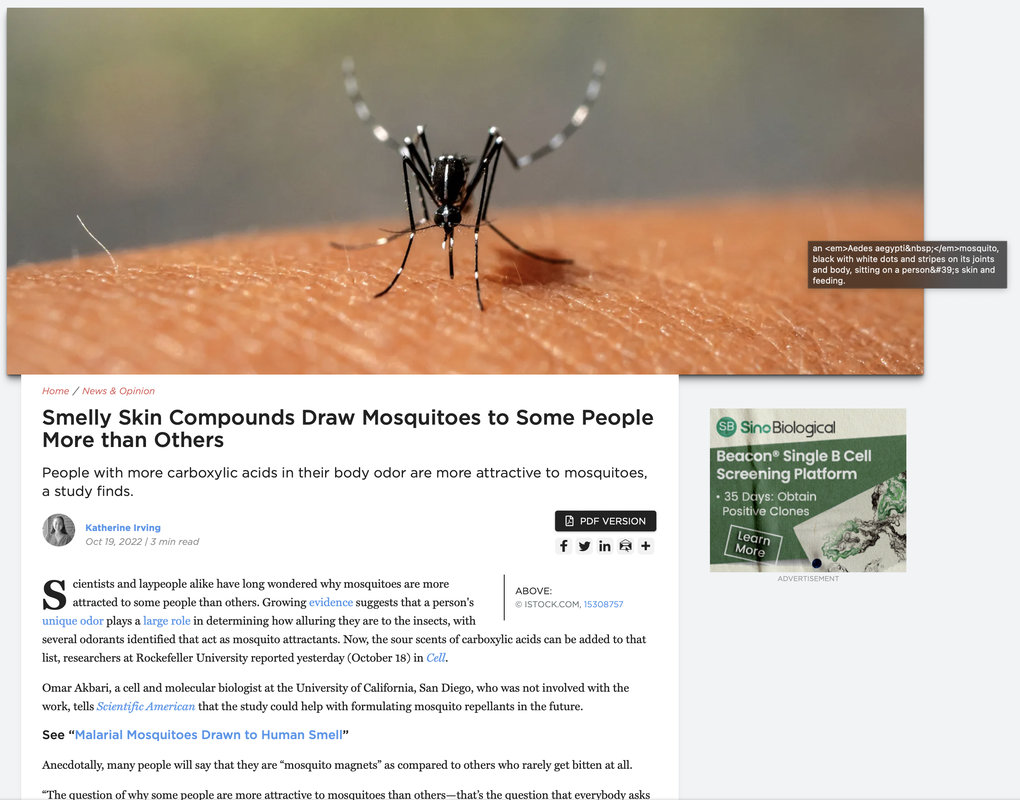
05 OCTOBER 2022By Dr Robyn Raban, Research Data Analyst, The Akbari Lab, University of California San Diego As part of its efforts to contribute to an informed debate on gene drive, the Outreach Network for Gene Drive Research conducted a series of video interviews with experts and researchers working on gene drive research. In one of these videos - “Why do we need to do field releases of gene drive technologies as part of the R&D process?” - Dr Mamadou Coulibaly of Target Malaria and I tackle the importance of field trials in gene drive research. Gene drive technologies have the potential to support the prevention of diseases such as malaria and dengue. For many years, gene drive technologies for vector control have been studied in the laboratory. However, these laboratory studies are not fully reflective of the conditions these technologies might eventually encounter in the field: field conditions are complex, highly variable and more stressful to gene drive organisms than those encountered in the laboratory. There are many variables in the field that can impact the success of a gene drive, such as for example:
“Why do we need to do field releases of gene drive technologies as part of the R&D process?” with Dr Robyn Raban, The Akbari Lab, and Dr Mamadou Coulibaly, Target Malaria. To learn more, you can watch the video here. These factors also vary over space (e.g., local versus landscape level) and time (e.g., seasonally). Therefore, while laboratory studies provide important safety and efficacy data needed to determine whether gene drive technologies could benefit disease control programs, they fall short of providing the necessary data to determine if these technologies are suitable for large scale use. The transition from the laboratory to the field is an essential step in the research and development process. If a gene drive technology were to be approved for field evaluation, early field trials would be conducted on a small scale to ensure performance and safety, before scaling to larger and more complex environments. Following a stepwise approach will help provide more reliable performance and safety data for stakeholders to assess whether a gene drive technology is effective and safe. It will also allow to collect data at each step of the research pathway, which in turn will help in building better predictive models of gene drive behavior on a large scale and ensure that these potentially life-saving technologies are developed in a responsible manner. |
Archives
June 2024
|


 RSS Feed
RSS Feed
